Patient Safety is our Priority
- At Next Wave India, we take our responsibility beyond just making medicines; we also focus on protecting the patients who use them. Through pharmacovigilance, we keep track of safety information constantly. This helps us identify even rare or unexpected adverse events.
- By sharing your experience, you help us improve medicine safety and protect patients everywhere.
Who Can Share Safety Information
- Patients, caregivers, healthcare professionals, pharmacists, distributors, and anyone who notices a safety issue with Next Wave India manufactured medicines can submit a report.
- Next Wave India encourages anyone who sees or experiences a possible adverse event, product quality problem, or unexpected reaction to share that information.
- You don’t need to be sure that the medicine caused the event. If you suspect there may be a connection, your report is still valuable.

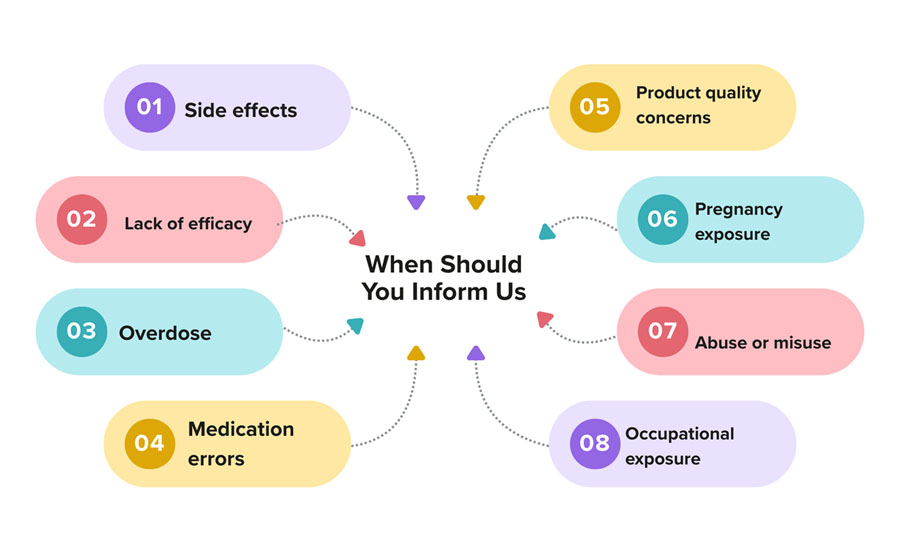
When Should You Inform Us?
- Please let us know as soon as you notice or suspect an adverse event after using our medicine. Consider reporting any unexpected side effects, lack of expected effectiveness, medication errors, product quality concerns, or unusual reactions after using the product.
- Early reporting helps us monitor medicine safety better and supports ongoing evaluation to ensure patient well-being.
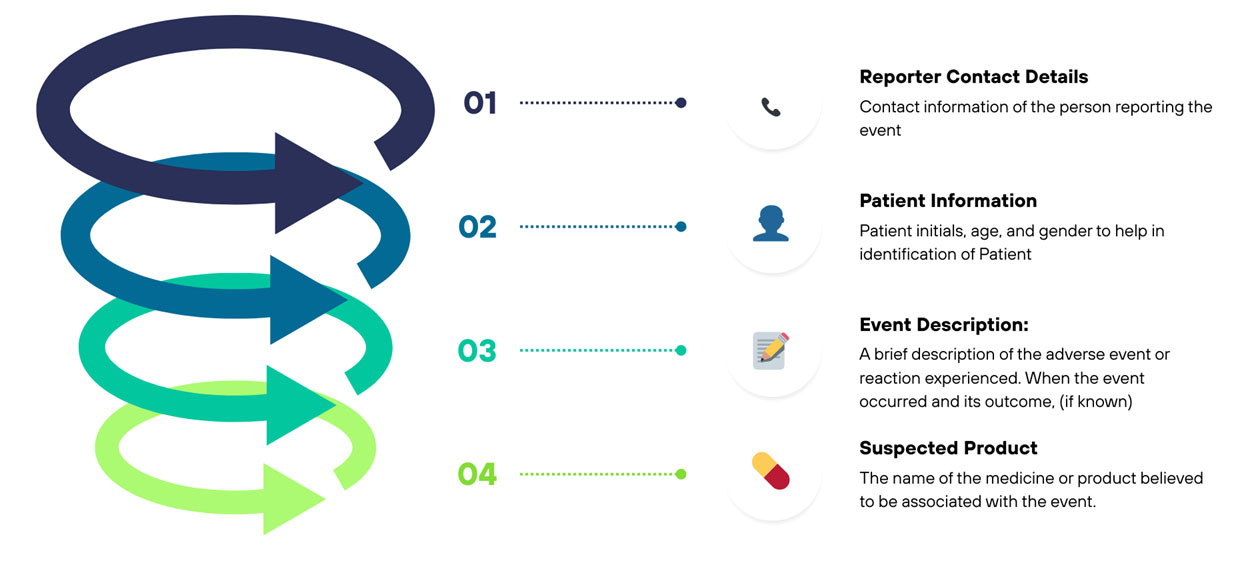
What Information Should You Share
- To assess a safety concern properly, it helps to provide key information such as patient initials, age, gender, the name of the suspected medicine, and a clear description of the adverse event. Details about when the event happened, the dosage taken, and any treatment received can also aid in the evaluation.
- If you have it, additional information like relevant medical history, other medicines being taken, laboratory reports, or medical documents can further help team understand the event and conduct a thorough safety assessment.
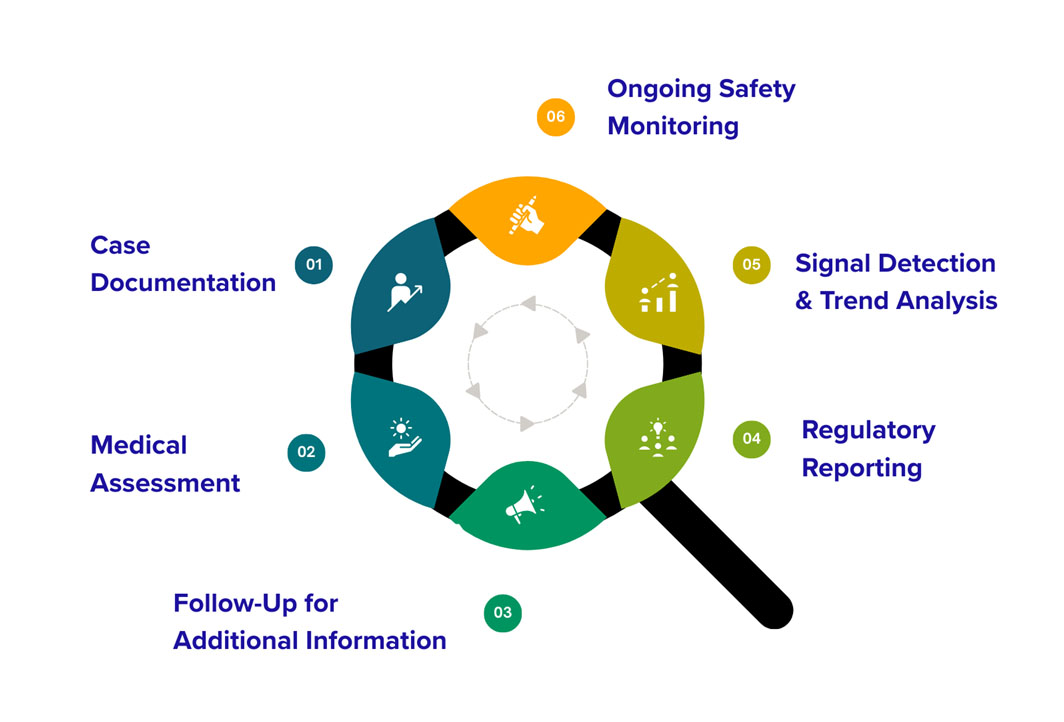
What Happens After Reporting?
- Case Documentation – Your report is securely recorded in our pharmacovigilance safety database for proper tracking and evaluation.
- Medical Assessment – Qualified pharmacovigilance and medical professionals review and assess the reported information.
- Follow-Up for Additional Information – If necessary, our team may contact the reporter to obtain further details that help improve the quality and completeness of the safety evaluation.
- Regulatory Reporting – If required, the case is submitted to relevant health authorities in accordance with regulatory timelines.
- Signal Detection & Trend Analysis – Reported information may be analyzed along with other safety data to identify potential safety signals or patterns.
- Ongoing Safety Monitoring – The information contributes to continuous safety monitoring to help identify potential risks and improve medicine safety.
Why Your Report Matters
- Every safety report is crucial for enhancing our understanding of how medicines work in real-world situations.
- By sharing your experience, you help us keep track of the safety and quality of our products.
- Your information can help identify rare or unexpected side effects, detect potential safety signals early, and support actions that strengthen patient protection. Even a single report can lead to safer medicines and better healthcare for patients around the world.

How & Where to Submit a Report
- 📧 Email: safety@nextwaveindia.com
- 🌐 Online Form: Available 24/7 on our website
- 📞 Helpline: +91-1704-223215 (Mon–Fri, 9:00 AM–6:00 PM)
- 📬 Postal Address: Rampur Ghat Road, Paonta Sahib, Distt. Sirmour, (H.P.)-173025, India
- You may also download the Adverse Event Reporting Form & submit it via email or post.
Go to Adverse Event Form
